
Immune System Reprogramming: Successes and Limitations of CAR-T Cell Therapy
Author: Xianhao Wu
May 08, 2026
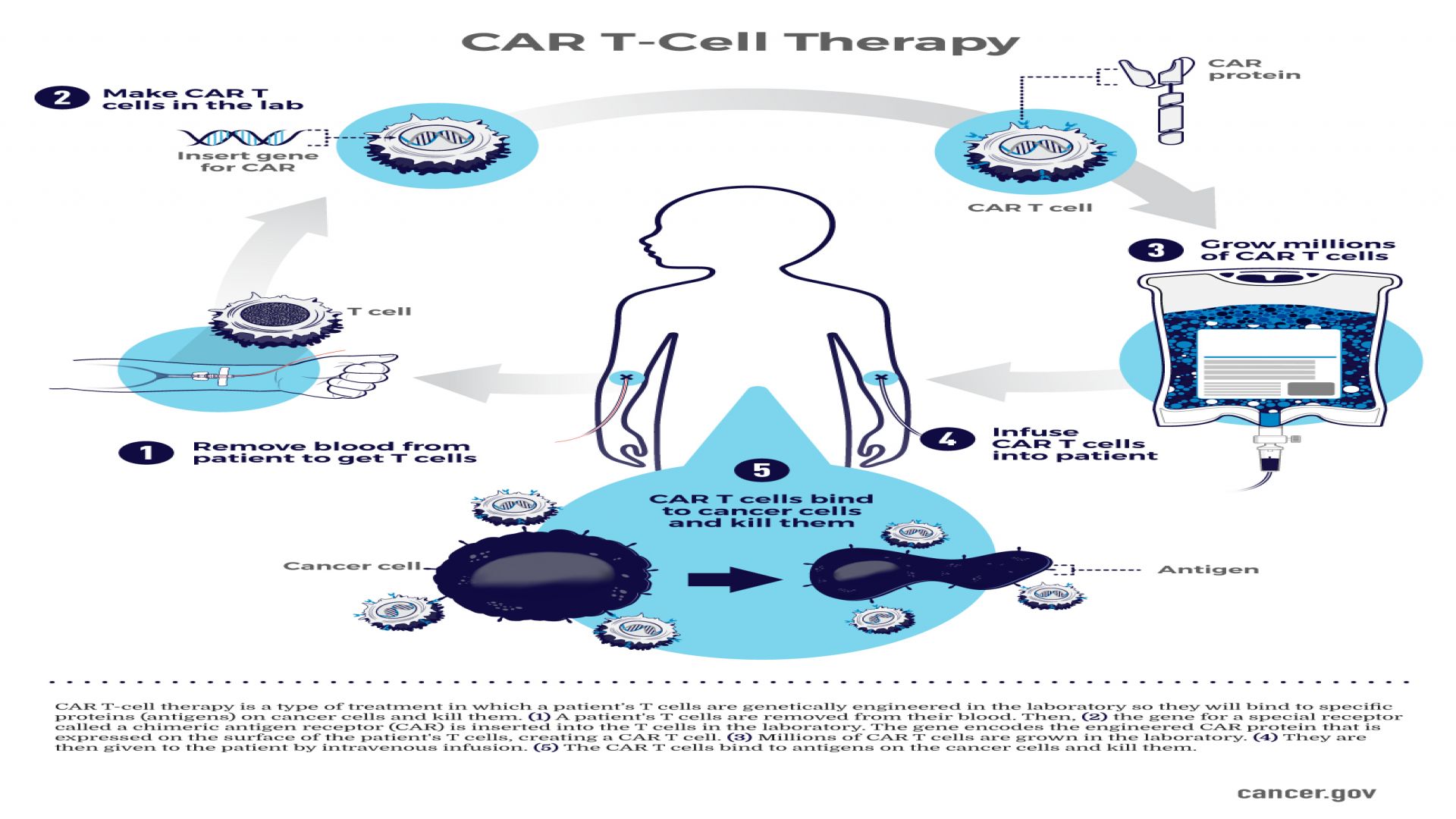
Traditional cancer treatments have saved countless lives, yet they often cause extreme side effects due to their lack of specificity in distinguishing cancer cells from healthy tissue (National Cancer Institute [NCI], 2022). Over the past decades, advances in genetics have reshaped how scientists understand cancer, leading to immunotherapies that target diseases more precisely (June et al., 2018). While CAR-T therapy has produced remarkable results for certain blood cancers since its 2017 FDA approval, its effectiveness remains limited by biological and practical challenges (NCI, 2022).
CAR-T cell therapy genetically reprograms a patient’s T cells, which are immune cells that patrol the body to eliminate infected or abnormal cells (Cleveland Clinic, 2024). Firstly, T cells are collected and extracted from the patient’s bloodstream through the process of leukapheresis (American Cancer Society [ACS], 2022). Then, a chimeric antigen receptor, or CAR gene, is inserted into these T cells using a viral vector (Newick et al, 2017). This gene directs the cells to produce a receptor protein on their surface that binds to a specific cancer antigen (Lim & June, 2017). Depending on the cancer antigen present, the CAR gene sequence is modified to alter the T-cell receptor proteins (Neelapu et al., 2018). After genetic modification, the CAR-T cells are grown in the laboratory and then reinfused into the patient (ACS, 2022).
By bypassing normal immune recognition pathways, CAR-T therapy demonstrates how gene therapy can significantly enhance the immune system's ability to target cancer (Newick et al, 2017). CAR-T cell therapy is particularly effective against blood cancers where tumor cells circulate freely and consistently express the same marker since the CAR T-cells are designed to recognize a single, well-defined antigen like CD19 (Maude et al., 2014). Patients with acute lymphoblastic leukemia and certain B-cell lymphomas have shown the greatest clinical success (Schuster et al., 2019). The ability of CAR-T therapy to specifically target cancer cells based on particular antigen expression and minimize harm to healthy tissue is one of its biggest benefits (June et al., 2018).
Since CAR T-cell therapy is most effective if the cancer consistently expresses one specific antigen, patients must undergo genetic and molecular testing before treatment (Majzner & Mackall, 2018). As a result, CAR-T therapy is not a universal solution, but a treatment whose success depends heavily on the biological characteristics of an individual patient’s tumor (Majzner & Mackall, 2018). Additionally, the therapy itself carries significant risks in terms of who can safely receive it (June et al., 2018). A major risk of CAR-T therapy is cytokine release syndrome (CRS) (ACS, 2022). This syndrome can cause multiple immune responses in the body, ranging from high fever, low blood pressure, organ dysfunction, and neurotoxicity (NCI, 2022).
Crucially, its risks and effectiveness depend heavily on a patient’s age, overall health, and access to care (June et al., 2018). Older patients or those with prior organ damage from earlier cancer treatments may face a higher risk of complications, reducing the overall benefit of CAR-T therapy (Cleveland Clinic, 2024). CAR-T therapy also requires weeks to manufacture, during which patients often need “bridging therapies” like chemotherapy to control disease progression, further limiting its use for individuals with rapidly advancing cancer (Cleveland Clinic, 2024).
Although many FDA-approved CAR-T treatments are covered by insurance in the US, coverage varies and out-of-pocket costs can be hundreds of thousands of dollars (NCI, 2022). Taking this into account, if one of my older aunts or uncles were diagnosed with B-cell lymphoma and was considering CAR-T therapy, I would only advocate for its use if they were able to tolerate bridging therapies during CAR-T cell production and if previous chemotherapy had not resulted in significant organ damage or immunological suppression.
The evolution of CAR-T therapy signals a broader shift in cancer treatment toward gene-based precision medicine (Neelapu et al., 2018). CAR-T therapy demonstrates how genetic engineering can treat cancer by directly reprogramming a patient's immune cells. Despite its current drawbacks, such as CRS, high cost, and decreased efficacy in solid tumors, ongoing research is quickly resolving these issues. Improved receptor design and safety "switches," two developments in gene-editing technology, are intended to lessen immune overactivation while extending CAR-T therapy to a greater variety of malignancies (Neelapu et al., 2018). Concurrently, the creation of commercially available CAR-T cells and combination treatments might reduce expenses and increase accessibility (June et al., 2018). Ultimately, CAR-T therapy is not only an effective cancer treatment, but also a fundamental model for the future of personalized medicine, where treatment is customized for the patient, the tumor, and the genomes.
References
American Cancer Society. (n.d.). CAR T-cell therapy and its side effects. https://www.cancer.org/cancer/managing-cancer/treatment-types/immunotherapy/car-t-cell.html
Cleveland Clinic. (2024, February 14). CAR T-Cell Therapy. https://my.clevelandclinic.org/health/treatments/17726-car-t-cell-therapy
June, C. H., O’Connor, R. S., Kawalekar, O. U., Ghassemi, S., & Milone, M. C. (2018). CAR T cell immunotherapy for human cancer. Science, 359(6382), 1361–1365. https://doi.org/10.1126/science.aar6711
National Cancer Institute. (2022). CAR T cells: Engineering patients’ immune cells to treat their cancers. U.S. Department of Health and Human Services, National Institutes of Health. https://www.cancer.gov/about-cancer/treatment/research/car-t-cells
Newick, K., O'Brien, S., Moon, E., Albelda, S. M. (2017). CAR T-cell therapy for solid tumors. Annual Review Medicine, 68, 139-152. https://doi.org/10.1146/annurev-med-062315-120245
Neelapu, S., Tummala, S., Kebriaei, P. et al. (2018). Chimeric antigen receptor T-cell therapy — Assessment and management of toxicities. Nature Reviews Clinical Oncology, 15, 47–62. https://doi.org/10.1038/nrclinonc.2017.148
Majzner, R. G., & Mackall, C. L. (2018). Tumor antigen eEscape from CAR T-cell therapy. Cancer Discovery, 8(10), 1219–1226. https://doi.org/10.1158/2159-8290.cd-18-0442
Maude, S. L., Frey, N., Shaw, P. A., Aplenc, R., Barrett, D. M., Bunin, N. J., Chew, A., Gonzalez, V. E., Zheng, Z., Lacey, S. F., Mahnke, Y. D., Melenhorst, J. J., Rheingold, S. R., Shen, A., Teachey, D. T., Levine, B. L., June, C. H., Porter, D. L., & Grupp, S. A. (2014). Chimeric antigen receptor T cells for sustained remissions in leukemia. New England Journal of Medicine, 371(16), 1507–1517. https://doi.org/10.1056/nejmoa1407222
Schuster, S. J., Bishop, M. R., Tam, C. S., Waller, E. K., Borchmann, P., McGuirk, J. P., Jäger, U., Jaglowski, S., Andreadis, C., Westin, J. R., Fleury, I., Bachanova, V., Foley, S. R., Ho, P. J., Mielke, S., Magenau, J. M., Holte, H., Pantano, S., Pacaud, L. B., . . . Maziarz, R. T. (2018). Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. New England Journal of Medicine, 380(1), 45–56. https://doi.org/10.1056/nejmoa1804980
Copyright © 2023 The Global Horizon 沪ICP备14003514号-6